A swift and accurate detection test is very important when it comes to battling a global pandemic like that of COVID-19. It allows the medical team to detect illness, support patients, and limit the spread of the disease. Currently, there are two major methods for testing COVID-19 – molecular test and antigen test. And now, researchers at Wellcome Sanger Institute have devised a new test called the INSIGHT that will allow patients to conduct COVID-19 test themselves and get preliminary result inside two hours.
 However, it could still be prone to false-negative and false-positive results. That is why the second step tests the validation of the results of the first test.
However, it could still be prone to false-negative and false-positive results. That is why the second step tests the validation of the results of the first test.
COVID-19: Current Testing Methods:
The two most popular tests for COVID-19 infection involves taking nasal swabs from patients. In molecular tests, researchers look for genetic materials of the virus, which when detected confirms the infection. On the other hand, in antigen tests, researchers look for viral proteins instead. Antigen tests are simpler in nature and can provide results faster but are less accurate than molecular tests. The problem with these tests is that they cannot be conducted at home. These tests require special equipment and are mostly conducted in laboratories by professional medical teams. Different research teams around the world are working on near-patient tests but they have not been that effective as the data collection is still centralized. There has always been some kind of delay between the test and its result.INSIGHT Test for COVID-19
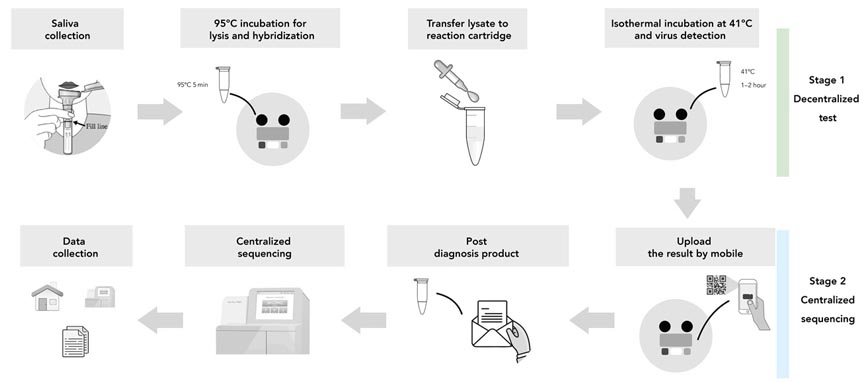
Researchers at Wellcome Sugar Institute have put forward INSIGHT as the possible solution for the present scenario. It makes use of both isothermal NASBA and Next Generation Sequencing techniques.Stage 1
The test is carried out in two steps among which the first is the isothermal NASBA (Nucleic Acid Sequence-based Amplification). During this phase, a sample of the patient's saliva is taken using a home-based kit at the point of care. According to the researchers, the isothermal NASBA reaction is able to amplify viral RNA to a detectable level within two hours. They have also claimed that the new test is as sensitive as the popular RT-PCR test but doesn’t require complex equipment.
Stage 2
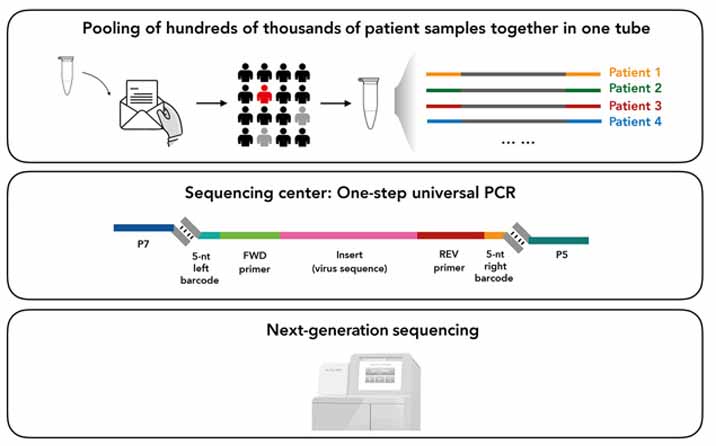
The second test reduces the risk of home testing errors. This process involves sending the sample to a laboratory by stamping a readable DNA barcode to the RNA sample. There, researchers will sequence tens of thousands of samples simultaneously to detect COVID-19 inside 12 hours.
Advantages
The new INSIGHT test for COVID-19 has numerous advantages over the RT-PCR test. First, taking a saliva sample is a lot easier than a nasal sample. The latter is difficult to carry out correctly and it can also be an uncomfortable experience for the patients. Thus, there is always a possibility of inaccurate results. Also, it doesn’t require complex tools to carry out. Researchers have proposed fluorescence and dipstick readout for the NASBA tests which they believe can be fitted inside a home-kit. INSIGHT method is also flexible. For instance, stage 1 and stage 2 can be used simultaneously or as independent tests on the basis of the availability of the equipment and testing laboratories.Moving Forward
Here, the INSIGHT test for COVID-19 is already a finalist in the XPRIZE Rapid COVID Testing competition with a prize pool of $6 million. The team of researchers is now working on patient samples for the validation of INSIGHT. After this, they will work on home-based kits to be used by patients at home.- Also, check out our review of the Xiaomi Mi 10i.
Article Last updated: February 14, 2021








